What do you picture when thinking of prescription drugs? Chances are you envision a translucent orange bottle filled with small oblong pills. Maybe you recall an asthma inhaler or a topical cream for treating eczema. Whatever the route of administration, the prescription becomes a part of your routine, at least for a time. It must for it to work in many cases.
What if it doesn’t have to be this way?
For most patients, the onus is on them to regulate their schedule of administering their prescription. Patients with chronic illnesses need to medicate throughout their lives, which for many is cost-prohibitive and subject to error. Between 40 and 50% of patients with chronic illnesses do not adhere to their prescriptions. Medical nonadherence is detrimental to the care of millions of patients. Which has biologists wondering…
What if we could program our cells to produce our prescriptions?
This science-fiction idea to turn our cells into custom pharmaceutical factories is not so far-fetched. The National Institute of Health unveiled a program to develop drug-producing cells in patients with clinical trials scheduled to begin within a decade. Biotechnology is developing personalized cellular pharmacies to produce therapeutic drugs to permanently treat, or perhaps cure, many chronic illnesses.
There are 3 main classes of drugs patients can receive.
Small molecules (i.e., chemicals)
Biologics (i.e., proteins)
Cell therapies
For most of the 20th century, small molecules were the only source of pharmaceutical research and development. These small molecules are so-called because compared to biological molecules (DNA, RNA, and proteins) they are much smaller in structure.
Small molecules found in prescription medicines are often synthetic, meaning they are not found in nature at all, and are instead synthesized via chemical reactions in a pharmaceutical lab. The benefit of using synthetic compounds is they can behave in novel ways. For instance, consider ibuprofen, sold under brand names Advil, Motrin, and Nurofen. It was initially developed as a treatment for rheumatoid arthritis but later found success as a mild over-the-counter pain reliever and fever reducer. Given its simple synthesis process and generic brands, it can be purchased for a few pennies per dose. Ibuprofen has successfully relieved the pain of millions of patients since its discovery in 1961. Prior to its discovery, patients either had to handle their pain on their own or take toxic and addictive opioids. Imagine relying on opioids for the flu!
The second class of drugs—biologics—began with insulin. Insulin became the first hormone discovered in 1921, the first protein to be sequenced in 1951, and the first recombinant protein therapy approved by the FDA in 1982. It subsequently kickstarted the biologic drug industry.
Recombinant protein production is where a DNA sequence that is not native to the organism is inserted into a cell to produce a particular protein it does not typically produce. Before 1982, insulin was sourced from the pancreas of dogs, pigs, and cows who each produce similar (but not the same!) insulin as humans. To overcome this, researchers took the DNA sequence for human insulin and inserted it into bacteria. The bacteria cultures began to produce human insulin from the DNA sequence the scientists inserted. Now, instead of harvesting the pancreas from living animals and purifying its insulin, scientists could instead culture bacteria in tubes and harvest human insulin directly from their culture.
Pharmaceutical companies already take advantage of cells to produce biologics in massive bioreactors: stainless steel egg-shaped vessels full of cells programmed with recombinant DNA to produce a therapeutic protein of interest. Yet there are limitations. Biologics are annoying to deal with. Their method of administration is limited to consistent blood injections. They also require constant refrigeration, throughout shipping and while in patient’s personal storage. This cold chain is a significant contributor to their increased cost relative to small molecules. Perhaps we could eliminate this hassle by removing the supply chain altogether…
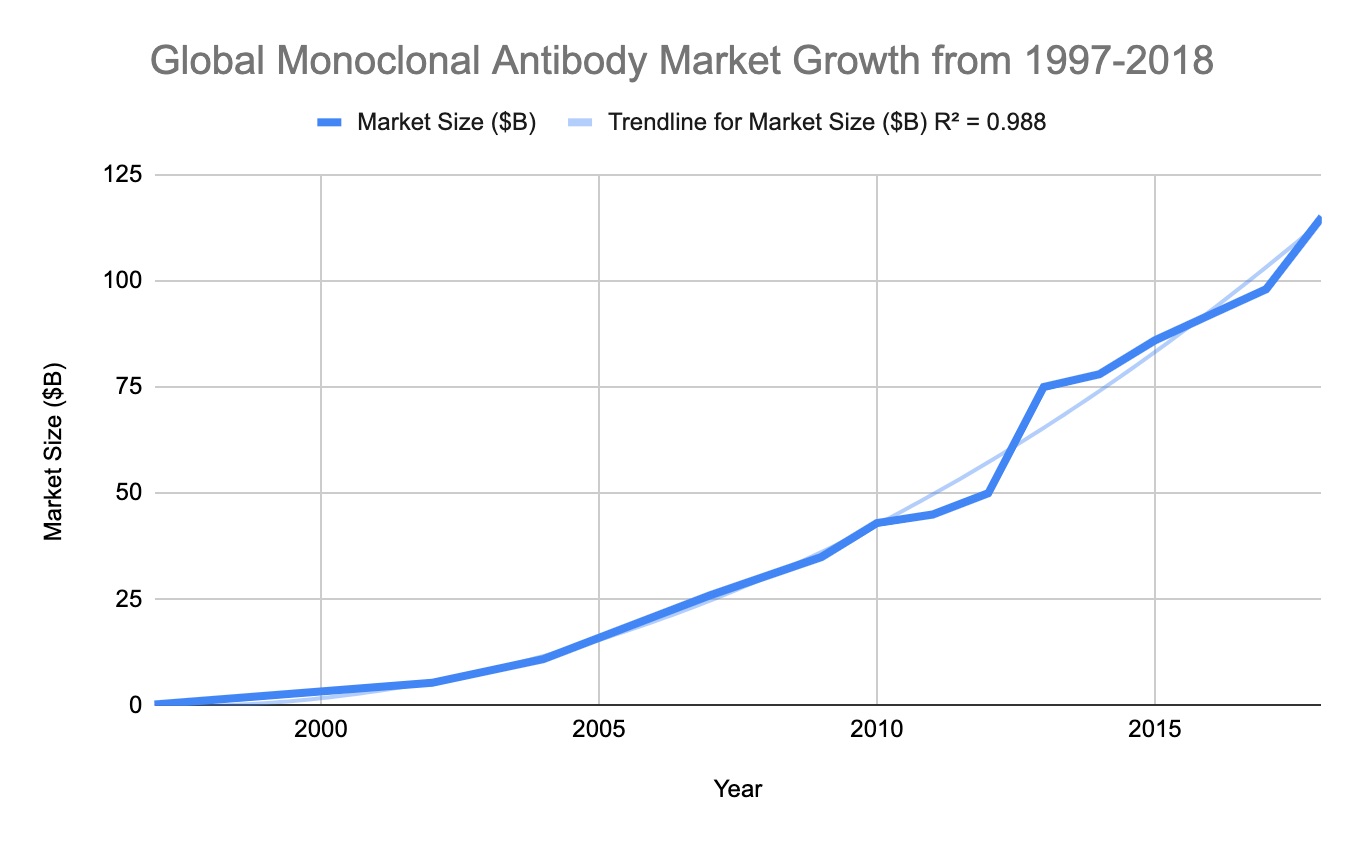
The next frontier of medicine, cell therapy, began in 2012. Six-year-old Emily Whitehead was diagnosed with acute lymphoblastic leukemia and after numerous rounds of chemotherapy the cancer had relapsed again. She was ineligible for a bone marrow transplant and her doctors told her parents there were no further options. The medical team recommended palliative care.
Emily and her family refused to give up, and the Whiteheads enrolled Emily in an experimental clinical trial at the Children’s Hospital of Philadelphia to do something truly transformative. Scientists extracted Emily’s immune cells, engineered them with a custom receptor to target her cancer cells, and injected them back into her bloodstream. The chimeric antigen receptor T cell (CAR T) therapy improved Emily’s death sentence condition within hours. In 2022, Emily and her family celebrated 10 years cancer-free. Her bravery in taking this experimental therapy has enabled hundreds of pediatric doctors to learn from her experience and treat thousands of patients around the world. It showed the world the potential of cell therapies.
Cell therapies, compared to small molecules or even protein biologics, are orders of magnitude more complex. We’re talking about using our cells, the building blocks of our being, to treat diseases. The limits are seemingly endless as we can engineer them with novel functionality, such as adding synthetic receptors to target a patient’s cancer cells à la CAR T therapy. This groundbreaking moment has inspired dozens of organizations and thousands of scientists (including myself!) to push the envelope of how far cellular therapies can go in treating cancer and many other diseases. CAR T cell therapies are currently in clinical trials for dozens of cancers as well as other diseases such as lupus, and other cell types are being investigated to treat chronic illnesses like ulcerative colitis and arthritis.
CAR T cells opened the door to engineer cells as a treatment for devastating diseases. Even the US government sees the potential for cellular therapies to transform the way we treat chronic illnesses.
The NIH unveiled the ARPA-H agency in 2022, a callback to the Department of Defense’s DARPA agency founded in 1958 to fund early research into world-changing technologies including the internet, personal computers, GPS, weather satellites, and Moderna’s COVID-19 vaccine. The goal is to inspire the same scale of technological breakthroughs defining the 20th century through DARPA into the biotechnology industry throughout the 21st. One of the first programs from ARPA-H is Resilient Extended Automatic Cell Therapies, or REACT. The REACT program is requesting proposals for how to implement a “Living Pharmacy”; engineered cells that following injection can be programmed to release a biologic therapy via an electronic device to control the dosage.
The idea in principle is simple.
Currently, pharmaceutical companies use large bioreactors full of cells programmed to produce human insulin. This insulin is harvested, purified, packaged, and sold to diabetes patients. The patient then monitors their blood glucose levels throughout the day and in particular, following meals. When they spike, they supplement their bloodstream with a dose of insulin by injecting it. Millions of diabetes patients rely on themselves to manually replace the function of a healthy pancreas every single day.
Instead, we could engineer cells to produce insulin within the patient when they receive an electrical signal from a mobile device. This would cut out the supply chain of producing, transporting, and storing insulin. It would also remove the need for daily injections to improve therapy adherence and patient quality of life.
We already engineer cells to produce therapeutic proteins in bioreactors. How do we electronically control the protein production of these engineered cells inside a person? As is often the case, nature has one idea for us.
Many bacteria are phototrophic, meaning they derive their energy from light via photosynthesis or a similar biochemical process. A lot of phototrophic bacteria derive all of their energy this way, so they are constantly searching for and harvesting light. For instance, phytoplankton occupy the upper surface of the oceans and fresh waters. These bacteria are so awash in light and so numerous that a byproduct of their energy production is our oxygenated atmosphere (more so than all plants combined!).
For bacteria in the deeper depths of these waters, the light and carbon dioxide needed for phototrophic energy production is far less plentiful. These organisms cannot rely solely on photosynthesis to provide their energy and must decide under different conditions which source to choose. One bacteria, Rhodobacter sphaeroides, is particularly efficient at determining which energy source to prioritize. These bacteria produce protein receptors called bacteriophytochromes on their cell surface to detect infrared light. These receptors control the expression of genes involved in photosynthesis. In the presence of light, the cells switch their energy source to phototropic processes; in the dark, they rely on chemotrophic processes where they harvest energy from molecules like sugars in their environment (“eating” their energy, like we do).
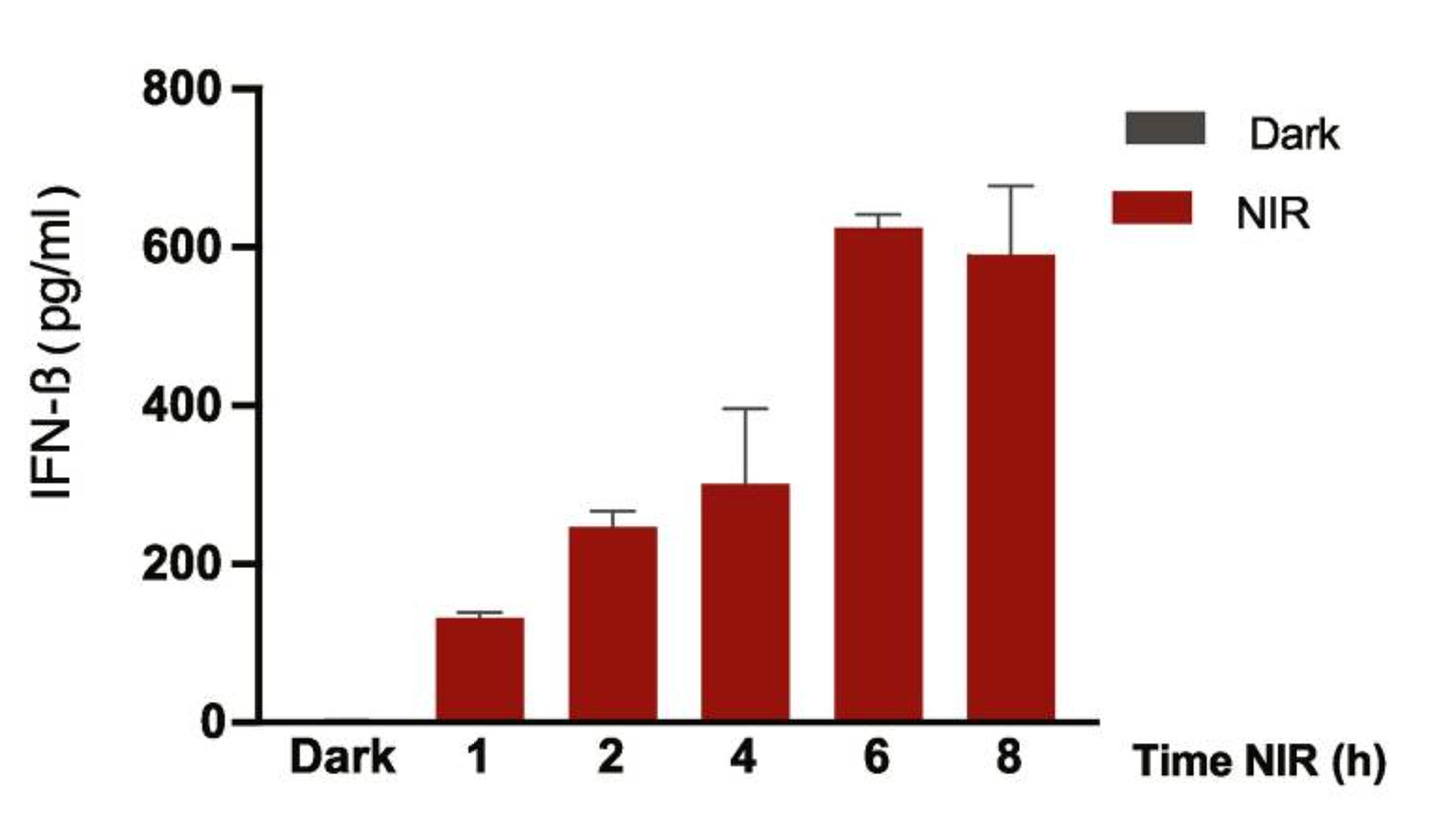
Researchers led by Marc Folcher at ETH Zurich utilized this process to engineer human cells to produce therapeutic proteins in the presence of infrared light from an LED. They took recombinant DNA encoding these bacteriophytochrome receptors and a therapeutic protein in response to their light detection. This recombinant DNA package was inserted into human cells. For the therapy, they chose interferon-β (IFN-β): a common treatment for multiple sclerosis that requires regular injections. When exposed to infrared light, the cells produced IFN-β, whereas in the dark the cells did not. They placed these cells on a small device with an infrared light controlled by a mobile operator such as a smartphone, and surgically implanted the device in a mouse model of multiple sclerosis. The device successfully prevented demyelination—a hallmark symptom of multiple sclerosis—in the mice with the device on and reduced inflammation in their central nervous system.
Let’s zoom out a bit. These researchers took light-sensitive proteins from a phototrophic bacteria, inserted them into human cells to produce a therapeutic protein, created an implantable device for the cells to deliver this protein in mice, and demonstrated the therapeutic benefit of this device in a multiple sclerosis model. The modality of the therapy did not change; IFN-β is still what ultimately treats the disease. The route of its administration, though, is vastly different. A single minimally invasive surgery replaces manually administered injections multiple times per week throughout a patient’s lifetime. That would be a huge win for patient quality of life, treatment adherence, and disease management.
This is but one way of achieving a living pharmacy device, and while successful in this animal study, it is far from guaranteed to work in people. There remain questions about how sensitive the device is to its input—for instance, how rapidly the cells start/stop producing the therapy when the light is switched on/off—and how controllable the dosage of the treatment is (can you turn the light to “high” to get twice as much of the drug as the “low” setting?). The potential is there, though, for a device such as the one made by Marc Folcher’s lab to create a personalized cellular pharmacy for lifelong treatment of chronic diseases.
Using our cells to produce our prescriptions in our bodies is borderline science fiction. Nevertheless, the concept of minimally invasive cellular pharmacies, if successful, will be in clinical trials this decade. At least, for diseases treatable with biologics.
Producing small molecules in cells is a bigger challenge. Given their synthetic nature and the fact that biology’s cells only produce natural products, there is no way for our cells to produce these molecules using natural processes. However, synthetic biologists and bioengineers are investigating novel enzymes to catalyze these reactions. Biocatalysis is entering the production of many small molecules as a more cost-effective, efficient, and accurate producer of many compounds when compared to traditional chemical synthesis. The future could see chemotherapy drugs produced within the tumor, eradicating the side effects associated with its systemic administration.
Cell therapy is not a replacement for existing small molecule or biologic treatments; it is a complement to unlock their full potential. In the same way that companies branching into biologics did not displace small molecules, cell therapies are a new tool to expand our ability to maximize the clinical benefit of existing therapies and uncover countless novel ones.
The move into recombinant and engineered biologics inspired by insulin in the 1980s led to a dramatic shift in pharmaceutical and biotechnology resources, towards complementing existing small molecule development with protein therapies. Now, with CAR T therapy’s breakthrough in pediatric lymphoma patients, the industry has added cell therapies as their next platform for treating disease. We are entering the next chapter of therapeutic development, one where we harness the power of our cells to combat disease and combine cell therapies, biologics, and small molecules into a single platform.