Liquid(ia) Gold $LQDA
This is Part 1 of a series. See Part 2 here and Part 3 here.
No, not that Liquid Gold!
The following is a very high risk stock. Please do your own due diligence and do not rely on this article for your research. I’m an investor, I get things wrong, please reference this post for more thoughts on why you should not rely on my work.
Okay, this post is about Liquidia Corp.
Overview
Liquidia Corp ($LQDA) is a $340M market cap, $261M enterprise value biotech company that manufactures drugs using their proprietary particle engineering system. They intend to commercialize their flagship product, Yutrepia, for the treatment of the orphan disease pulmonary arterial hypertension (PAH), a $5B market in the United States. PAH is a fatal disease characterized by increased pressure in the pulmonary vasculature and that often results in heart failure.
Unlike many publicly traded biotech companies, Liquidia faces zero clinical trial risk. Yutrepia is tentatively approved by the Food and Drug Administration (FDA), which means that the drug is almost ready to launch. To receive final approval from the FDA and launch the drug, Liquidia needs to successfully resolve the outstanding patent infringement claims levied against them by United Therapeutics ($UTHR), the leader in the PAH space.
Despite recent material wins on the patent litigation front, the share price of Liquidia implies a low probability of their legal success. With shares trading at $5.42 I estimate the market is implying a 36% chance that Liquidia prevails; I think the odds are much closer to 75% which justify a $13.70 share price today (+153%). If Liquidia is successful and launches Yutrepia then the shares are worth ~$18 (+232%) at launch. If Liquidia is unsuccessful, the shares are likely to collapse, probably to the vicinity of $1 (-82%). I repeat, this is a high risk stock.
Brief Company Backgrounds
Liquidia’s biggest competitor is the market leader, United Therapeutics (UTHR), whose flagship product, Tyvaso, delivers patients treprostinil through a nebulizer. UTHR’s market cap is $10B and the company generates revenue of $1.8B per year, 43% from the sale of inhaled treprostinil and 90% from the sale of PAH treatments more broadly (UTHR sells infused, oral and inhaled PAH medicines).
Martine Rothblatt, motivated by her daughter’s PAH diagnosis, founded United Therapeutics in 1996. Rothblatt, a lawyer by training and founder of SiriusXM radio, brought on Roger Jeffs, a Ph.D. in pharmacology, in 1998 as Director of Research, Development and Medical of UTHR. Together, Rothblatt and Jeffs grew United Therapeutics into a PAH powerhouse by their decision to commercialize treprostinil in 2002. Today, treprostinil is sold under the brand names Remodulin for infusion, Orenitram for oral, and Tyvaso for inhalation. All three brand names are owned by United Therapeutics.
After an 18 year career, Jeffs retired from UTHR as co-CEO (a title he shared with Rothblatt) and President. In January of 2022, Jeffs became CEO of Liquidia. Jeffs owns 2.7% of Liquidia’s shares outstanding, approximately $10M worth.
The Patent Infringement Cases
United Therapeutics sued Liquidia for infringement of three of their patents: U.S. Patent Numbers 9,604,901 (the 901 patent), 9,593,066 (066), and 10,716,793 (793). By suing Liquidia for patent infringement UTHR guaranteed that, even if they lose in court, Yutrepia’s launch would be delayed by several years due to the time it takes to try and appeal the various court decisions.
Before the lawsuit, Liquidia was a promising drug company targeting an established, multi-billion dollar market with a superior drug-delivery mechanism. Today, Liquidia’s stock is a bet on the outcome of patent litigation. Similar to when an investment grade bond is downgraded and holders have to automatically sell, this lawsuit triggered indiscriminate selling amongst LQDA holders and the share price has languished ever since. This lawsuit is why the opportunity exists.
Liquidia can launch Yutrepia if all asserted patent claims are found to be either invalid or not infringed. So far, Liquidia has definitively won on two out of the three patents (901 and 066), and has scored a major victory on the third patent (793).
Definitive Victories
The 901 patent is a product by process patent for the preparation of treprostinil, the ingredient in UTHR’s Remodulin. In June 2021, during a claim construction hearing for Hatch-Waxman litigation, UTHR agreed to the entry of judgment of Liqudia’s non-infringement of the 901 patent. The 901 patent is no longer a roadblock for Liquidia – they won.
The 066 patent is a product by process patent for the preparation of treprostinil, the active ingredient in Remodulin, relating to UTHR’s product Tyvaso, a nebulized solution for the treatment of PAH. In August 2022, the same court in the Hatch-Waxman litigation mentioned above found that all 066 patent claims asserted have been found to be either invalid or not infringed. Liquidia won on the 066 patent.
Victory in the Making?
Two down, one to go. Liquidia has one more hurdle to clear: the 793 patent which covers treprostinil administration by inhalation. This patent was issued by the US Patent and Trademark Office (PTO) in the month following UTHR’s filing of the lawsuit. Despite the delayed timing, UTHR was able to add the 793 patent to the proceedings and make it a gating factor for Liquidia.
The 793 patent is also where the story becomes slightly more confusing. Liquidia made two separate cases for the invalidity of the 793 patent: one in front of the Patent Trial and Appeal Board (PTAB) of the PTO in an Inter Partes Review (IPR) and one in front of the district court as part of the Hatch-Waxman litigation.
Liquidia was successful in invalidating the 793 patent in front of the PTAB at the IPR. This is a hugely important point. While not sufficient to end the patent issue today, it is a necessary step on the way to victory. Let’s leave the IPR hearing and go back to the Hatch-Waxman litigation for a second.
Unfortunately for Liquidia, they were not successful in front of the district court in the Hatch-Waxman (HW) litigation for the 793 patent. Why? Because Liquidia was estopped, or precluded from making the same argument in both the HW and IPR proceedings. Liquidia decided to use their strongest case in the IPR and won. By default, the HW case was decidedly weaker and Liquidia lost.
What does it all mean, and how does Liquidia move forward from here? The most straightforward way for Liquidia to prevail on the 793 patent is to have the PTAB’s IPR decision affirmed on appeal. This process takes time and will likely conclude sometime in 2023/2024 (more on the timeline later). Once this occurs, the 793 patent will be thrown out, the district court’s ruling will be irrelevant, and Liquidia will launch Yutrepia.
A second avenue for Liquidia to get to market is to launch Yutrepia “at-risk.” Today, they are precluded from doing that due to the HW ruling. However, Liquidia has asked the judge who ruled in HW to effectively look at the big picture. Liquidia’s rationale, among other things, is that they have already invalidated the 793 patent – the only patent stopping them from launch – in the PTAB and are just waiting on the decision to be affirmed on appeal which happens 73% of the time. Liquidia thinks they should be able to launch now and face the consequences of the appeal process later. If Liquidia launches now and wins on appeal at a future date, then no harm, no foul. If Liquidia launches now and loses on appeal at a future date, then they owe damages to United Therapeutics. This outcome makes the most sense to me, but I am not a lawyer and don’t want confirmation bias to enter my thought process. To be clear, Liquidia’s lawyers are presenting a far more sophisticated legal argument than what I just described. Because it is a sophisticated legal argument I am not qualified to handicap its chances of success. Accordingly, I consider the prospect of a favorable judgment a “free option” that could payoff as soon as this month.
Timeline
Standing between Liquidia and launch are the following steps: The PTAB rehearing request being denied, an appeal filed, record transmittal, briefing, oral arguments, and a decision. To bracket the timeline I think the shortest case is an August 2023 launch and the longest case is in-line with Liquidia’s guidance of a mid-2024 launch. The key variables are 1) how fast the rehearing request is denied and 2) whether or not Liquidia receives a Rule 36 decision. On point 1, the PTAB aims to decide on rehearing cases within a month of receiving a request. UTHR waited until the very last day – consistent with running out the clock on a weak case – in mid-August. One month from that date has already past, but it’s possible that the rehearing request is denied this month, October 2022. The worst case I have seen is a 9 month lag between a rehearing request and a decision from the PTAB – this puts approximately 7 months between best/worst case on the PTAB decision. On point 2, if Liquidia receives a Rule 36 decision, that shaves 3 months off the decision timeline. Otherwise, a Full Written Decision would take 3 months. Altogether, best case is August 2023 and worst case is June 2024. Liquidia guided the market to “mid-2024 or earlier” which is conservative and the best decision; better to underpromise and overdeliver.
Why I think Liquidia Receives a Rule 36 Decision
Federal Circuit Rule 36 specifies that “the court may enter a judgment of affirmance without opinion” when certain specified conditions exist. A Rule 36 Decision “does not endorse or reject any specific part of the trial court’s reasoning” and is non-precedential, i.e. not binding on the court. These judgments are usually handed down after the court hears oral arguments. What does this mean? It means the timeline to affirm the PTAB’s decision on appeal can be dramatically shortened by a Rule 36 Decision. I think Liquidia is likely to receive one due to the precendent set by United Therapeutics in their eerily similar case vs. SteadyMed. SteadyMed filed an IPR for one of UTHR’s patents which also happened to be a process to prepare treprostinil, the active ingredient in Remodulin. Long story short, the patent was found to be invalid by the PTAB, and received a Rule 36 Decision when it was affirmed on appeal. Liquidia’s case is so close to SteadyMed’s that one might think UTHR is running the same exact legal playbook to run out the clock in the face of an inevitable defeat. Note: when the clock ran out, UTHR ultimately acquired SteadyMed.
What’s it Worth?
What’s it worth? I will better flesh out the math in a future post. Let’s assume LQDA is able to capture 8% of the PAH market (PAH WHO Groups 1 and 3) by 2028. In that scenario, I estimate the company is able to generate $1B in revenue. At a peer multiple of 3.2x EV/Sales, discounted back to today at 10%, I arrive at a share price of $34. This seems like a reasonable bull case and assumes a lot of execution success.
If you’d rather run napkin math, consider the following. UTHR is worth about $10B. What’s a post-clinical, imminently-commercial competitor worth, arguably one with a better product and led by a pioneer of the industry? I’d say 10% of UTHR is fair. That gets you to ~$1B or $18/sh.
Today the market implies Liquidia has a 36% chance of prevailing in court. I think the odds are much closer to 75% which justify a $13.70 share price today (+153%). If Liquidia is successful and can launch Yutrepia then I think the shares will re-price to $18 (+232%) in short order. Longer term, Liquidia has the potential to materially penetrate the PAH market and be worth in excess of $30. If Liquidia is unsuccessful, the shares are likely to collapse to the vicinity of $1 (-82%).
Risks
Liquidia is facing a formidable challenger. Martine Rothblatt’s identity is tied up with the success of United Therapeutics. By all accounts, Rothblatt does not like to lose. Expect UTHR to fight Liquida tooth-and-nail here. As mentioned several times already, the downside to the stock is substantial (-82%) if Liquidia loses in court.
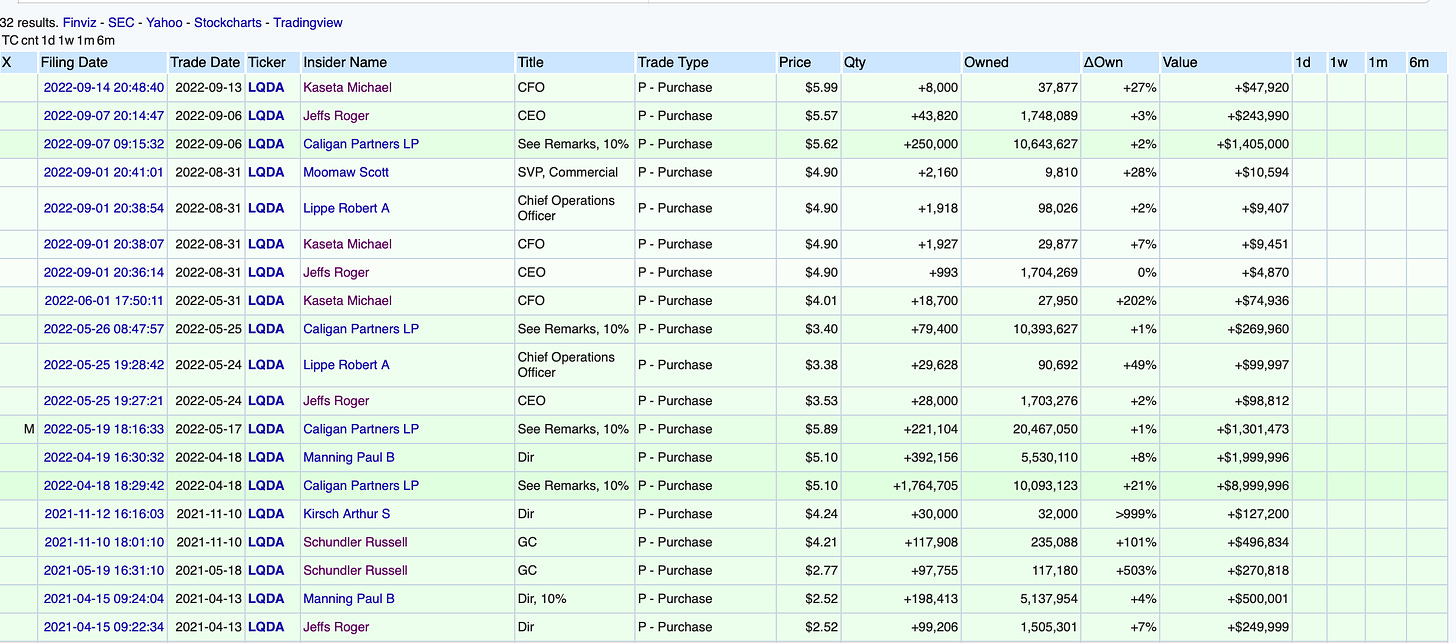
A Note on Insider Ownership
The Hatch-Waxman decision was handed down at the end of August and insiders started buying almost immediately. Insiders had already been buying ahead of the HW decision. CEO Roger Jeffs owns ~$10M of stock. Fund Management Company and Board Member Caligan Partners owns ~25% of the company! Yes, they bought after the HW concluded, despite Liquidia’s loss on the 793 patent.
This is Part 1 of a series. See Part 2 here.