I need to level with my readers about something. Drug names? They can be hard to pronounce. This is one of the many fine reasons that pharmaceutical companies come up with snappy brand names. This drug, the subjective today's column, has one of the snappiest brand names of all time. Abilify. I just want to Abilify everything. I want to be Abilified. It's truly a shiny example of modern pharmaceutical branding. One of the things that makes it such a great name is that the actual name of the drug is difficult to pronounce from scratch. So everybody refers to Abilify as Abilify, and not by its generic name. Game? Won.
This is one of the many medications in the pantheon of psychiatric treatments that I have had the pleasure of taking in the past. I gained about 60 pounds on this medicine! That problem is not unique to me, and it features prominently in the story of Abilify. It was a blockbuster drug for Otsuka Pharmaceuticals, at its peak, bringing in around $6.9 billion in annual sales.
Susequent articles in this series cover Abilify in Bipolar Mania and Abilify in Bipolar Depression.
Oh, hello there! Welcome to The Frontier Psychiatrists. It’s a daily newsletter about health. I have a whole series about psychiatric medications, and this is the next installment. As a programming note, most of my posts are free at first. But I write a lot, and after a few weeks, articles get plugged behind a paywall. It’s well over 500 posts by this point—a silly book or three on healthcare, pharmacology, the history of medicine, building health businesses, and the like. I strongly suggest subscribing, it encourages me to do more of this, so you don’t have to work out all these details by yourself. I benefit from your encouragement, and my paid subscribers are very encouraging.
Abilify is a “second-generation antipsychotic,” otherwise known as an “atypical antipsychotic.” It has already made guest appearances in prior articles about CATIE as it was included in that comparative effectiveness trial in schizophrenia. This article is going to be forced to limit itself, however. The Abilify story is just too big.
It’s revenue gives us a hint of why…

The drug’s FDA indications include:
Schizophrenia (14.1)
Acute Treatment of Manic and Mixed Episodes Associated with Bipolar I
(14.2)
Adjunctive Treatment of Major Depressive Disorder (14.3)
Irritability Associated with Autistic Disorder (14.4)
Treatment of Tourette’s disorder (14.5)
The injection is indicated for:
Agitation associated with schizophrenia or bipolar mania (14.6)
Regardless, that is a lot of work to do the research needed for all those indications and probably would require an article each to do them justice. Right now, I’m not going to bother to do that.
This is a lot of indications for any one drug. Honestly, I think, in retrospect, this speaks well of the team at Otsuka—they didn’t just promote it off-label illegally and pay the fines later like Gabapentin. Oh, wait.—they also promoted it illegally according to the $4m settlement with DOJ.
The TL;DR
I’m going to save readers time, to the best of my ability, which has, of course, been abilified.
Schizophrenia: Yes, ok, fine.
Bipolar Disorder: No, but I will deal with it in another article.
Adjunctive Treatment of Depression: Hell No (as covered just the other day).
Irritability associated with Autism: Nope, and I will address it below.
Tourette: Eh, I will review this in another article later.
(injectable for) Agitation: I don’t see the point. We have other options.
Pharmacokinetics and Mechanisms… Worth Mentioning!
Abilify binds to dopamine D2 receptors—a mechanism that, in schizophrenia, has come under some scrutiny recently1—but does so as a partial agonist. It’s not just “blocking” the receptor. At low dopamine concentrations, it acts as an agonist. At high concentrations, it acts to block the receptor and is thus a partial antagonist. The binding affinity, however, is absurdly high—it sticks to the dopamine receptor—like glue—and will displace most other drugs. It also has a very long half-life (about 75 hours) and an active metabolite (dehydro-aripiprazole) with a 94h half-life.2
Abilify sticks around in your system, for a long time. This makes it ideal in case someone “abruptly stops” because it tapers itself, more or less, no matter what you do. There are now several long-acting injectable formulations of aripiprazole that last six weeks or more per dose. I think these long-acting injectable versions are the best use of the drug, and I’ve written on the topic myself.3
Schizophrenia: It’s Better than Placebo
Thank god I spent all the time I did teaching my readers to read Forest Plots in meta-analysis figures in prior articles like this one and this one.
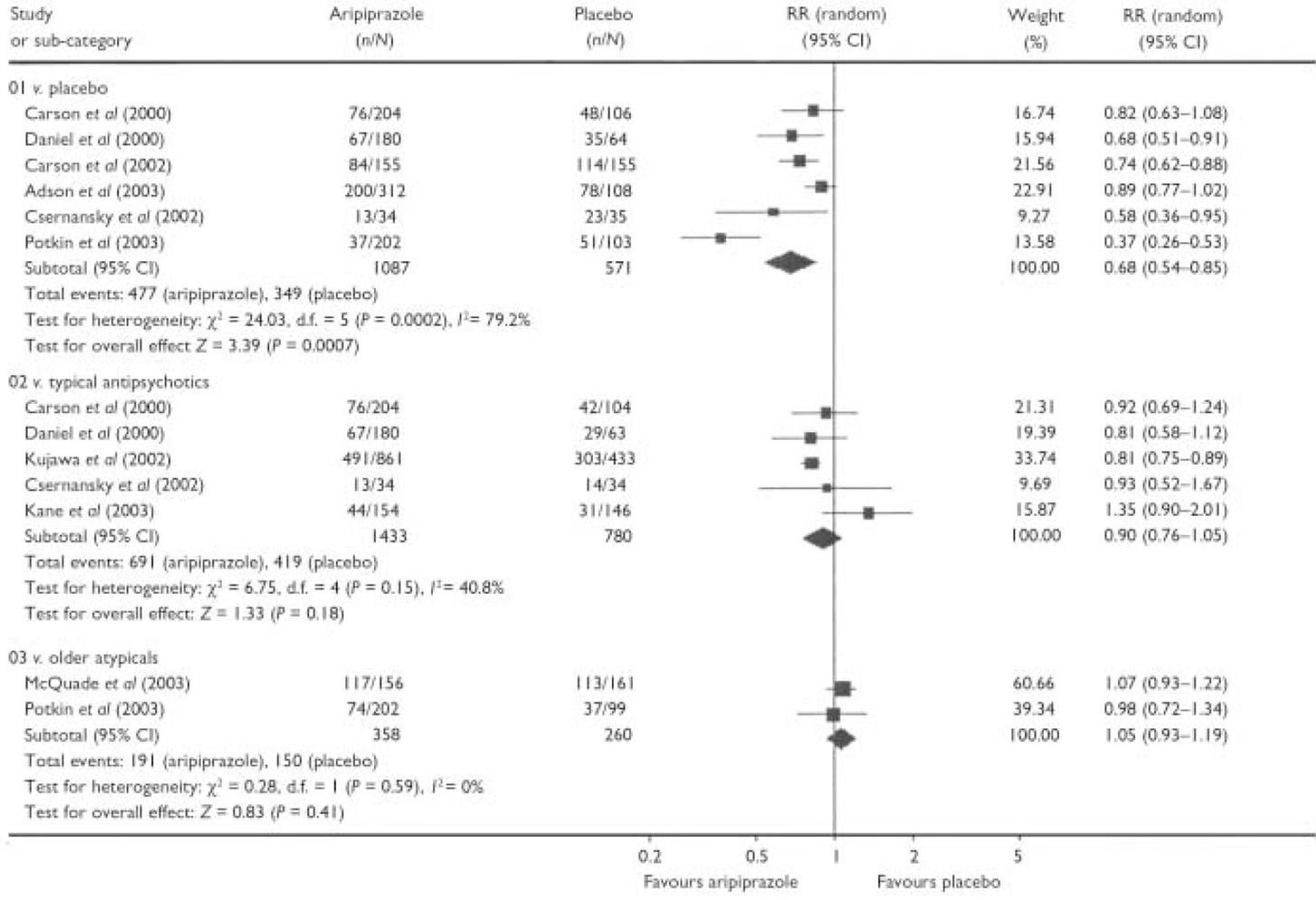
Schizophrenia treatment is difficult to study because the patients have psychosis AND good sense. What I mean by that is when the medicine isn’t helpful, even psychotic individuals will drop out of the study, and this has caused serious methodological issues in research on schizophrenia, as reviewed in this article about the use of aripiprazole.4
Medication adherence is a real problem in all of medicine. Even in cancer treatment, patients are not great at taking their oral medicine regularly (an issue I discuss with the brilliant Anthony Sterns, Ph.D. of iRxReminder in this podcast). In schizophrenia, the adherence rate is about 50%.5
In a research trial, evaluators need to decide what to do about “dropouts.” This is a major issue in aripiprazole trials, historically (El-Sayeh, Et. al.):
7 of the 10 studies that were included reported data in terms of both a last-observation-carried-forward (LOCF) analysis and an observed-cases analysis (where observed cases are defined as those who completed the trial). We could not use the LOCF data because of the high drop-out rates reported in the studies as well as the tendency to report mean figures without providing a measure of variance6.
There is another statistical method that one could use, and I strongly prefer, called using a mixed-effects model. However, as a non-statistician, the short version is when you have high drop-out rates, sometimes there is only so much one can do. So many people bail on the trials it makes it hard to understand what do to with the data.
The issue with only analyzing individuals who complete the study is called “survivorship bias.” Suffice it to say, it’s a problem. Abilify works in schizophrenia, but not for everyone.
Irritability Associated with Autism…Problematic Data.
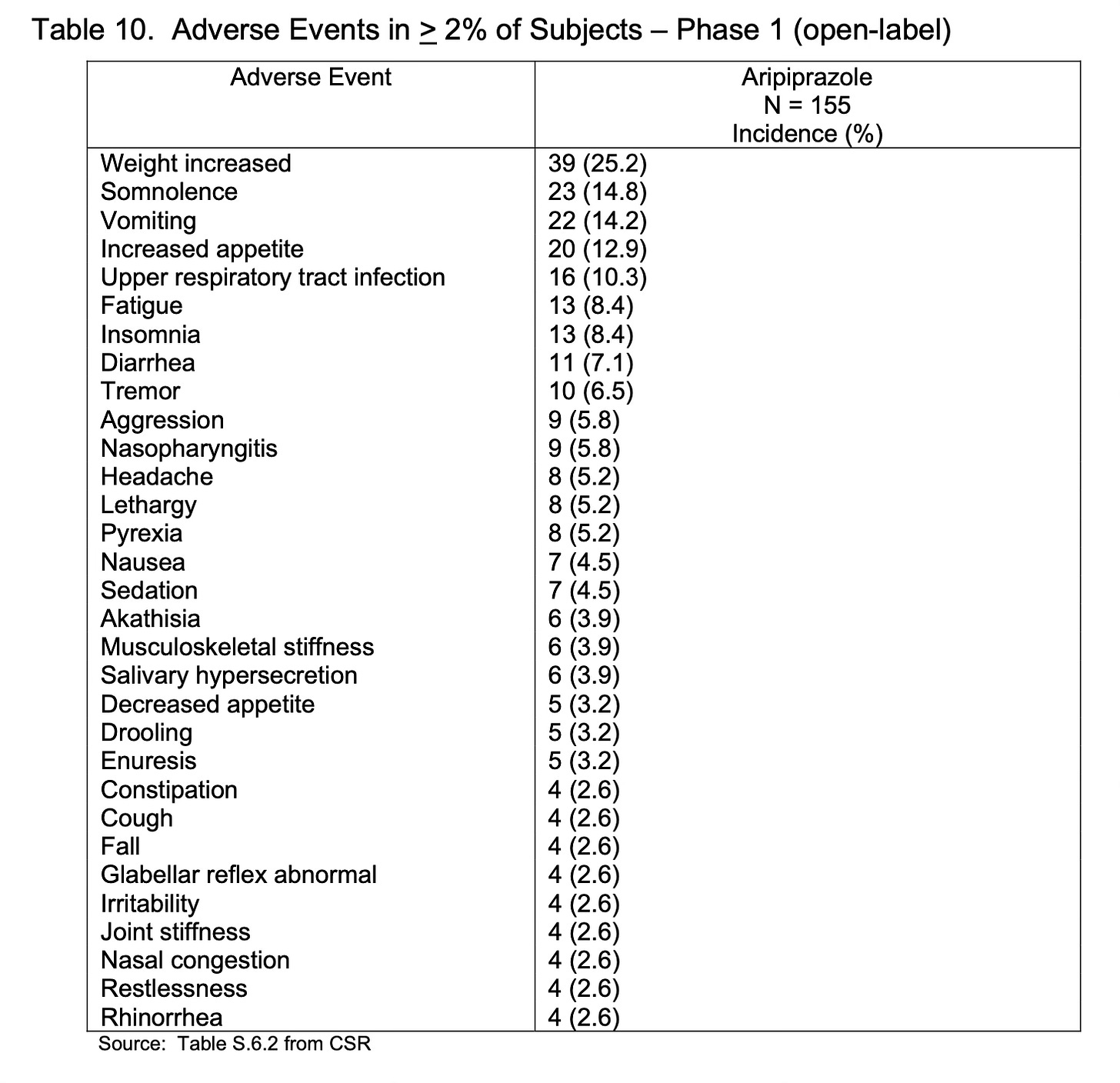
Although the initial FDA approval trials established the superiority of Abilify to a placebo in the acute treatment of irritability in pediatric patients with autism, this effect was not sustained, and the FDA declined to provide a label for “maintenance” of this acute treatment given the negative study results submitted by Otsuka for the indication. This result is especially notable given the long-term risks of aripiprazole including weight gain in 25% of irritable, autistic kids:
Frankly, it’s kind of amazing and somewhat horrifying that medical education doesn’t require just a year or more of reading through FDA filings on the medications we are learning to manage. As an aside, the genre of “dry, regulatory sarcasm” is wildly underrated. Just take a moment with a gem such as the following (from page 25):
“This reviewer had a difficult time trying to determine whether this additional analysis was ever performed.”
The FDA’s review of Abilify in this indication has just a hint of exhaustion in its dry, regulatory tone:
It is unlikely that the results from this clinical trial would significantly alter the clinical practice of prescribers. Patients would continue to be monitored and, if irritability symptoms continue to show improvement with aripiprazole, it is likely that aripiprazole would be continued in that individual patient.
The FDA review of this non-label makes it clear—people are going to prescribe this anyway. Nothing we say about this negative trial will stop them.
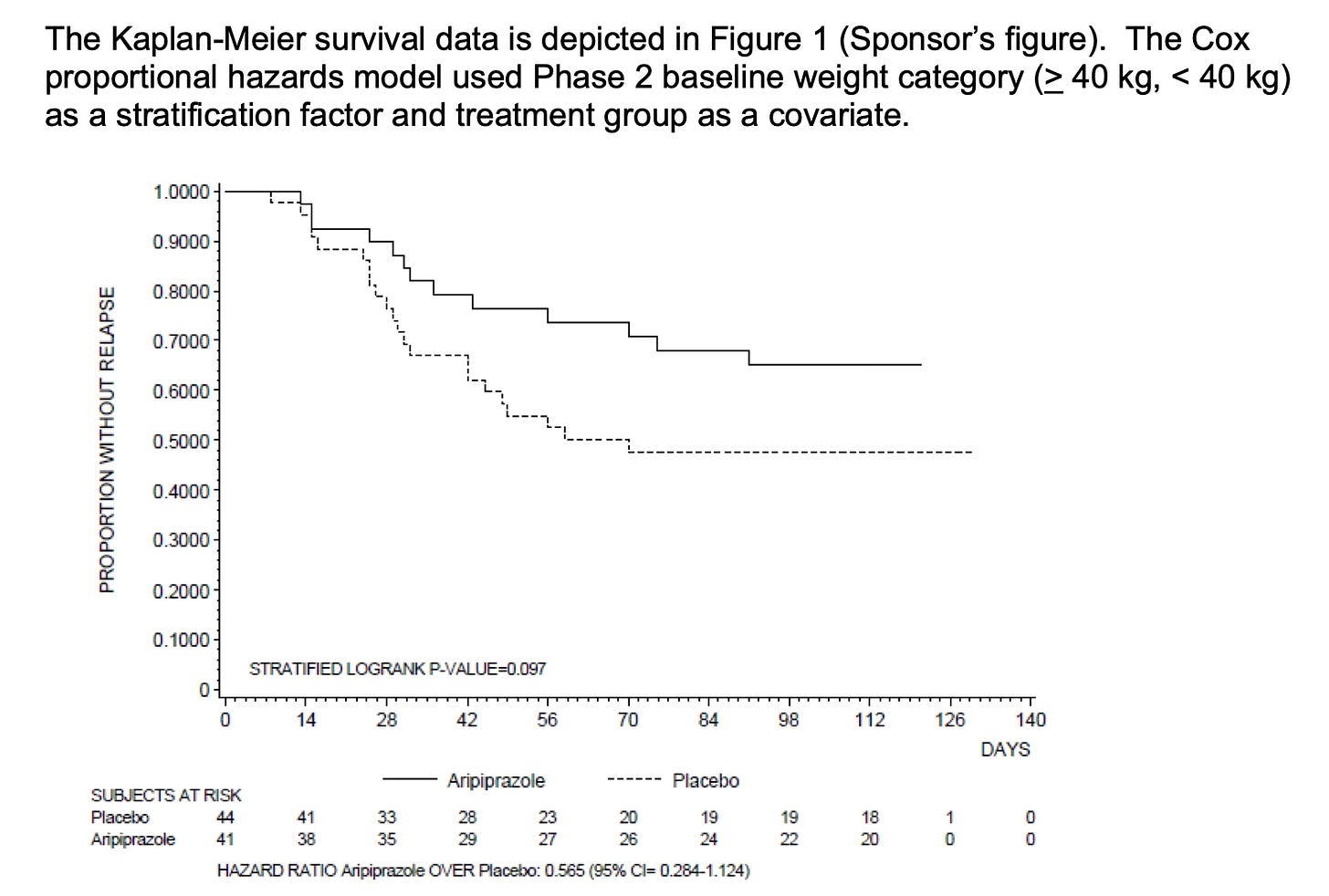
Even when it doesn’t beat placebo over the long term:
See that “P-Value of 0.097”? That isn’t significant. That is what a negative trial looks like. Here is—and I’m not kidding—the FDA has as its proposed non-labeling complete with weird redactions:
It is at this point that I mention: that youth treated with high-dose antipsychotic medicines in Medicaid populations die more often than those not treated with these drugs, at a rate 500% greater than dying by fentanyl overdose. Thus the lack of sustained benefit in this indication is all the more concerning. It might work acutely, but please, don’t keep kids on it, I implore my colleagues. To quote the FDA, there is cognitive dissonance about discontinuing these medicines:
Although not impossible, it does not seem likely that a drug that shows benefit in the acute treatment phase would not be effective in the maintenance treatment of the disorder.
That is one day's worth of Abilify writing. I felt it imprudent to go on and on about this one drug in one day’s article.
Prior articles about antipsychotic medicines include great reasons to subscribe to this amazing resource for your life and or practice:
Risperdal (with a podcast version)
Clozapine, Part 1.
A Review of Tardive Dyskinesia
Finally, some of my own journey with obesity treatment after antipsychotic treatment.
Thanks for reading, and consider becoming a paid subscriber, and sharing it your friends and enemies. They need engaging educational content, too.
Yun S, Yang B, Anair JD, Martin MM, Fleps SW, Pamukcu A, Yeh NH, Contractor A, Kennedy A, Parker JG. Antipsychotic drug efficacy correlates with the modulation of D1 rather than D2 receptor-expressing striatal projection neurons. Nat Neurosci. 2023 Aug;26(8):1417-1428. doi: 10.1038/s41593-023-01390-9. Epub 2023 Jul 13. PMID: 37443282; PMCID: PMC10842629.
Kim JR, Seo HB, Cho JY, Kang DH, Kim YK, Bahk WM, Yu KS, Shin SG, Kwon JS, Jang IJ. Population pharmacokinetic modelling of aripiprazole and its active metabolite, dehydroaripiprazole, in psychiatric patients. Br J Clin Pharmacol. 2008 Dec;66(6):802-10. doi: 10.1111/j.1365-2125.2008.03223.x. PMID: 19032724; PMCID: PMC2675764.
Correll, C. U., Muir, O., Al-Jadiri, A., Kapoor, S., Carella, M., Sheridan, E., ... & Kane, J. (2013, December). Attitudes of children and adolescents and their caregivers towards long-acting injectable antipsychotics in a cohort of youth initiating oral antipsychotic treatment. In Neuropsychopharmacology(Vol. 38, pp. S280-S280). MACMILLAN BUILDING, 4 CRINAN ST, LONDON N1 9XW, ENGLAND: NATURE PUBLISHING GROUP.
El-Sayeh, H. G., Morganti, C., & Adams, C. E. (2006). Aripiprazole for schizophrenia: systematic review. The British Journal of Psychiatry, 189(2), 102-108.
Acosta FJ, Hernández JL, Pereira J, Herrera J, Rodríguez CJ. Medication adherence in schizophrenia. World J Psychiatry. 2012 Oct 22;2(5):74-82. doi: 10.5498/wjp.v2.i5.74. PMID: 24175171; PMCID: PMC3782179.
One brief note to ambitious meta-analysis authors of the future: JUST CONTACT THE AUTHORS to get the unpublished variance data. We will, usually, be happy to provide it to you.



Just started my autistic 10 year old on aripiprazole today, about 2 years after it was first recommended. It seemed like the only alternatives were risperidone and Lamotrigine. It seems like we’ve tried everything. SSRIs, anti-anxiety meds, therapy, therapeutic school setting, completely reworking our parenting. Her behavior can be extreme and I worry that she’s going to get herself arrested or killed. I’ve already lost track of how many encounters we’ve had with the police. If you’re saying it’s a “nope”, what else would you suggest?
Was on this med for about 3 weeks. Did nothing for me, and by nothing I mean: if I were in a clinical trial I woulda thought I was in the placebo group. Not often I get to say a psychiatric med didn't even have the decency to give me a headache as a side effect.
I was given 5mg diazepam 3x a day with aripiprazole because the latter could be "activating". Due to all the anti-benzo campaigns in the UK I tried to avoid taking the diazepam because "diazepam is bad". After the fireplace started talking to me I was swapped onto 600mg quetiapine, and 18 months of side effect hell.