Part 2: ACUTE VIRAL RHINITIS - from Common Cold to Deadly Disease
PART 2
ACUTE VIRAL RHINITIS - from Common Cold to Deadly Disease
INTRODUCTION
The entry of the ‘novel’ Wuhan coronavirus (SARS COV-2) into the world viral microbiome has had several far-reaching consequences. Although infection with this virus may have been no worse than severe influenza (1, 2, 3), there is now universal recognition of the potential for a viral upper respiratory tract infection (URTI) to develop into a severe and sometimes fatal disease. Thus, the common cold and influenza have now become conditions that both patients and healthcare practitioners take more seriously – especially in vulnerable populations (i.e., the elderly and those with co-morbidities, such as obesity, diabetes and heart disease).
Research into therapeutic interventions that are focused on a specific virus may not prove fruitful, as the main factor in determining the clinical features and progression in an URTI is not the virus itself, but the response of the host’s immune system. Moreover, interventions that target one single pathway in the complex and staged host immune response, are likely to have unforeseen consequences, e.g., rofecoxib and other ‘anti-inflammatory’ drugs, (4, 5). This is most likely due to the dual nature of human immunity: the same cellular agents and chermical pathways that are activated to defend the host and clear the pathogen when there is an infection, also maintain homeostasis under normal conditions. Therefore, if a single pathway is suppressed in an attempt to control localised ‘inflammation’, other parts of the body may suffer. So far there is no single drug that is capable of preventing or stopping the progression of a viral UTI (including Covid-19). Part of the reason for this may be due to the fact that a single-targeted drug cannot address a disease which is a complex multi-factorial process. (6).
ACUTE VIRAL RHINITIS - WESTERN BIOMEDICAL PERSPECTIVE
Viral infiltration and infection
Acute viral rhinitis (AVR), a.k.a. the common cold, is mostly caused by human rhinoviruses. Other causative agents include coronavirus, respiratory syncytial virus and influenza viruses. Viral infection of the nasal epithelium occurs after inoculation through respiratory droplets or aerosols from an infected person. In general, children experience 8–12 colds a year, while adults usually experience 2–3 colds per year.
The hallmark symptoms of AVR are nasal stuffiness and discharge, sneezing, headache, sore throat, and cough. The common cold is usually a self-limited illness, confined to the upper respiratory tract. However, in some patients the infection spreads to adjacent tissues and organs, giving rise to different clinical features. Occasionally AVR predisposes patients to bacterial infections in the bronchi and lungs. Despite the usually benign nature of the illness, the common cold is an enormous economic burden on society in terms of visits to health-care practitioners, treatments, and absences from work, school, or day care. Every year, in the USA, about 25 million people visit their family doctors with uncomplicated upper respiratory infections, and the common cold syndrome results in about 20 million days of absence from work and 22 million days of absence from school. (7, 8, 9)
Host barriers to infection
The human respiratory system has evolved a complex series of barriers to repel potential invaders. In the nose, the mucosa and local lymphoid tissues constitute the main physical, immune and chemical barriers to viral infiltration and infection. These consist of the respiratory epithelium with its ciliated cells and overlying layers of mucous and microbiota, together with lymphoid cells both mobile and static, mostly residing within the lamina propria, together with their chemical secretions. The local lymphoid tissues, comprising the adenoid, bilateral tubule, palatine and lingual tonsils (collectively referred to as Waldeyer’s ring), play an important role in the adaptive immune response, which is specific to the infecting virus; while the resident lymphoid cells (macrophages, dendritic cells, killer T cells, memory T and B cells, natural killer cells and innate lymphoid cells) are the key players in the innate immune response, which is general and non-specific. The structure and function of the nasal mucosa and lymphoid tissues are described in my previous post.
Viral infection pathways – overcoming the mucus barrier
Once a virus has escaped the physical, immune and chemical barrier mechanisms within the nasal mucous layer, it is able to infect the nasal epithelial cells. The nasal microbiota and mucous layer provide a very effective barrier against the entry of bacteria and fungi. However, because of their extremely small size, viruses may readily pass through these layers and infect the epithelial cells, causing damage to tight junctions, disruption of cell membranes and eventually cell death.
The nasal mucus layer should present, in theory, an impassable barrier to viruses for two main reasons. Firstly because of the high density of this liquid (containing glycoproteins, antimicrobial proteins, immunoglobulins, etc.), a virus particle would take several days to reach the epithelial layer by passive diffusion alone, during which time it would have been cleared away, along with the mucus. Moreover, the glycoproteins within the mucus layer have specific sites which bind to the surface of virions (viruses), trapping them and further slowing the passive diffusion process, facilitating clearance.
The recent development of cryo-electron microscopy has allowed the building of reliable molecular models of entire viruses, in some cases at a nearly atomic resolution level. Based on the model of Influenza A virus, it has been proposed that viruses responsible for respiratory infections have a filamentous structure. The viral filament is able to rapidly traverse the nasal mucus layer by means of the receptor binding and cleaving proteins which are concentrated together at each pole of the viral filament. When one end becomes attached to a strand of glycoprotein, the other end is randomly moved around by Brownian motion. When the unattached end is brought into contact with a glycoprotein strand that is closer to the epithelium, it then binds with the deeper level glycoprotein and releases the other end. Thus, the virion is able to move in a ratchet-like manner through the mucous layer, making use of the host’s own barrier mechanism to attach to and then detach from adjacent strands of glycoprotein. (9, 10, 11)
Viral entry into epithelial cells
The infection process begins with viral entry into the nasal epithelial cells through binding to certain receptors on their apical surface. These receptors normally ‘sense’ noxious agents by temporarily binding to molecular stimuli from within the nasal passages. These molecular stimuli include molecules on the surface of micro-organisms or molecules which are the products of host cell destruction. Activation of these receptors leads to secretion of cytokines and chemokines which directly act on the cells of the immune system, priming the immune response and initiating local inflammation. Unfortunately, some of these receptors become binding sites for viral infiltration, whereby the virus is able to gain access into the cell. Once inside the cell, the virus is able to trigger an increase in the number of these specific receptors, thus increasing viral access sites on the cell surface. Once the cell has been infiltrated with viruses, this is followed by expression and duplication of viral genetic material. However, the cell soon dies because its metabolism has been taken over by the virus. Cell death after viral invasion allows free viruses to be taken up by immune surveillance cells which then migrate to local lymphoid tissues to initiate the host’s adaptive immune response. When responding to a virus with which the host’s immune system is already familiar (i.e. a new exposure to a virus that has infected the host previously), the immune activity mostly occurs within the nasal mucosa as memory cells are activated and the first tier of the local immune response is triggered. However, when exposed to a novel virus, the immune activity is more concentrated within the local lymphoid tissues, i.e. adenoids and tonsils. Thus, there are local as well as adjacent immune and inflammatory responses aimed at pathogen clearance and repair to damaged epithelial tissue. Importantly, the inflammatory response – and not the damage caused by the virus - is mostly responsible for the clinical features. (11, 12, 13)
Effects of viral infiltration on the nasal microbiota
The nasal microbiota is affected in many ways by viral infection. The best available evidence indicates that an acute viral infection of the nose allows a relative increase in the number and proportion of pathogenic bacteria in the local microbiome, which in turn facilitate the viral infection by directly activating the virus as well as disrupting the host’s innate immune responses. This points to a symbiotic relationship between viruses responsible for AVR and pathogenic bacteria that cause more severe respiratory infections such as pneumonia. (14, 15)
Immunological barriers to viral penetration
Beginning within the mucus layer, immune surveillance cells (macrophages and dendritic cells) are present in the deeper, more aqueous region that surrounds the cilia of the epithelial cells. In their quiescent state, macrophages clear away particulates, apoptotic cells and cellular debris to maintain homeostatic tissue function. The quiescent state of immune surveillance and immune cellular activity is maintained by constant chemical signalling from the epithelial cells. Once the virus has infiltrated to this level, epithelial cells, macrophages and dendritic cells are able to sense its presence through recognition of foreign molecular patterns on the surface of the virus. A complex series of chemical signalling takes place through which all three types of cells become activated by direct chemical stimulation and, in the case of the immune cells, by down-regulation or removal of epithelial cell quiescence signalling.
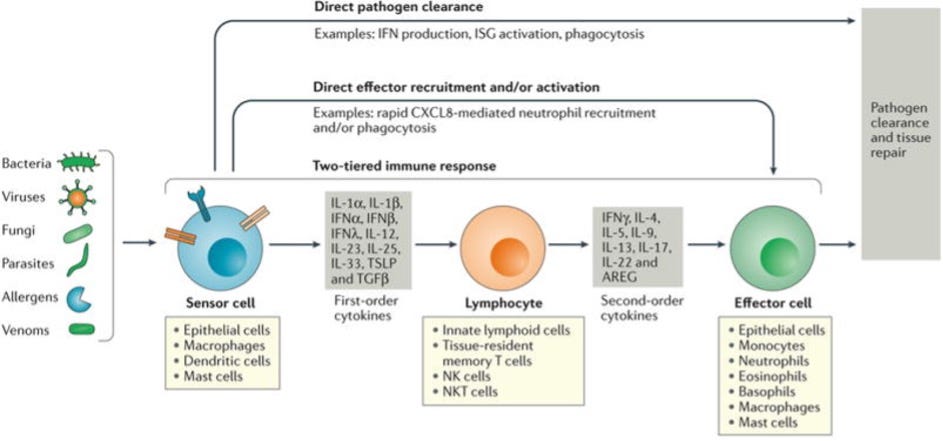
Once these immune sensor cells, including the epithelial cells, have been activated, they respond in two ways: a) by directly attacking and neutralising the virus and b) by recruiting other cells in either a single tier or two-tiered immune response (see Figures 1 and 2).
All of these responses are mediated by cytokines (including interferons), chemokines, anti-microbial proteins and growth factors, which are secreted by the sensor cells. Cytokines activate and regulate defensive cellular activity, while chemokines attract other immune cells and promote their movement into the local area (referred to as ‘recruitment).
In the single tier response, cytokines from the sensor cells directly activate and recruit effector cells that clear pathogens. These effector cells are mostly neutrophil leukocytes and monocytes. In addition, chemical signalling between the sensor cells also serves to promote their own phagocytic, cytotoxic and anti-viral activity. If this response fails to contain the viral infection, the two-tiered response is activated.
The in the two-tiered response, the sensor cells direct their chemical signalling through first-order cytokines, to activate the local resident lymphocytes: neutrophils macrophages, killer T cells, memory T and B cells, natural killer cells and innate lymphoid cells. This initial step is aimed at direct pathogen clearance, through phagocytosis (by neutrophils and macrophages), lysis, or binding of microorganisms (through the release of various proteolytic enzymes and anti-microbial peptides). Additionally, the resident lymphocytes may activate the specific response pathways of adaptive immunity if the virus has managed to escape the innate immune response.
The second tier of the local innate immune response activates a different set of local immune cells. The local lymphocytes, primed by first order cytokines, secrete second order cytokines that act upon several types of effector cells, mostly neutrophils, monocytes and macrophages, to complete the process of pathogen clearance and promote repair of damaged epithelial tissue. Within this staged response mechanism there are regulatory pathways to inhibit activation of subsequent stages once the pathogen has been adequately contained, thus minimising the inflammatory response, and limiting potential tissue damage. (16, 17)
FIGURE 1. Stepwise engagement of tiered responses following respiratory infection
Pathogens and certain noxious compounds are detected by sensor cells located within the respiratory tract. In viral infections, the sensor cells (epithelial cells, dendritic cells and macrophages) immediately initiate innate immune responses that may be sufficient to clear localized infections. For example, sensor cells may secrete factors such as interferons (IFNs) that lead to pathogen clearance (direct pathogen clearance, top arrow). In some cases, first order cytokines directly recruit effector cells that clear pathogens, e.g. recruitment and activation of neutrophils to clear pathogens (‘direct effector recruitment and/or activation’ pathway, second arrow from the top).
In addition to the direct and one tiered pathways, a two-tiered response can be engaged. Sensor cells secrete first order cytokines that act on tissue-resident lymphoid cell populations, which integrate these signals, destroying infected cells and releasing appropriate second order cytokines. These second-order cytokines, in turn, recruit and activate effector cells and effector functions specific to the pathogen type (e.g. with allergens the effector cells are predominantly mast cells, basophils and eosinophils, while with viral infections neutrophils, monocytes and macrophages are predominant). The effector cells serve to promote pathogen clearance and tissue repair. (16)
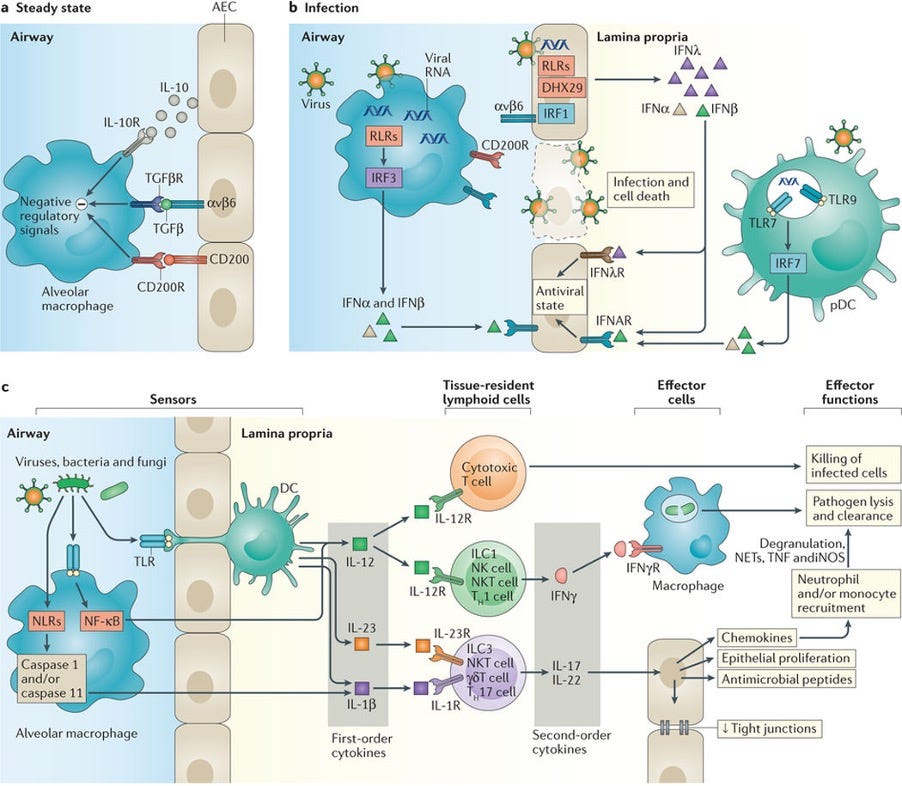
Figure 2. Single and two-tiered responses in type 1 immunity.
a. At steady state in the airways, alveolar macrophage activation is suppressed by negative regulatory signals from epithelial cells
b. During infection, disruption of these interactions due to death of epithelial cells enables activation of macrophages. Recognition of viruses by airway epithelial cells (AECs) leads to chemical signalling that induces an antiviral state in proximal AECs.
c. Local macrophages and DCs that extend trans-epithelial processes enable the recognition of viral, fungal, and bacterial molecules, leading to the production of first order cytokines. These first order cytokines act on tissue-resident lymphoid populations of cytotoxic T lymphocytes to enhance direct killing of infected cells, and on innate lymphoid cells (ILCs), natural killer (NK) cells, NK T cells, and T cells to induce the production of appropriate second order cytokines. These second order cytokines in turn act on AECs to induce chemokine production, antimicrobial peptide release, and increased proliferation and/or tight junction formation to enhance epithelial integrity and constrain pathogen spread. Local and chemokine-recruited phagocytes including neutrophils and monocytes are additionally activated by chemokines, enhancing their phagocytic capabilities and leading to enhanced pathogen lysis and clearance.
The danger of excessive immune response
Some types of cytokines released by epithelial and immune cells, e.g., interleukin-6, (IL-6) and tumour necrosis factor-alpha (TNF-a), have strong pro-inflammatory effects and are markers of disease severity. Other first tier cytokines stimulate the production, differentiation, proliferation, and survival of neutrophils (single tier response), as well as other cells that are involved in both first and second tier localised immune response.
Chemokines produced by these cells attract both innate and adaptive immune cells to the local area. If excessive or prolonged, the release of cytokines and chemokines by respiratory epithelial and other cells may lead to excessive leukocyte infiltration and activity, which results in excessive damage to respiratory tissue, e.g., in the lungs, leading to the development of acute respiratory distress syndrome. This process is referred to as ‘cytokine storm’. (6, 17)
Dendritic cells and macrophages
Both cell types are derived from the same progenitor cell line in the bone marrow and circulate in the blood as monocytes (which mostly develop into macrophages) and immature dendritic cells. Monocytes migrate into body tissues and differentiate into macrophages and may also, under conditions of inflammation, develop into mature dendritic cells (DC). In their quiescent state, macrophages clear away particulates, apoptotic cells and cellular debris; they are monitored and regulated mainly by the epithelial cells.
Local DC’s are derived from both immature DC’s in the circulating blood and also from monocytes that have been attracted to the local area by inflammatory signalling. Located within the lamina propria below the epithelium, DC’s extend trans-epithelial processes that sense viral infiltration in the deep mucous layer surrounding the apices of the epithelial cells. Once activated they initiate, coordinate, and regulate adaptive immune responses through their ability to capture and process viral antigens, migrate to the local lymphoid tissues and present these antigens to resident T-cells. Thus, they are uniquely able to induce naïve T cell activation and effector differentiation within the local lymphoid tissue. They are, in a similar manner, involved in the induction and maintenance of immune tolerance (i.e. suppression of immune activation and inflammation to non-harmful substances and micro-organisms) under normal homeostatic conditions.
Additionally, DC’s can secrete cytokines and growth factors that modify ongoing immune responses, and are, in turn, also chemically influenced by other immune cells, such as natural killer cells and innate lymphoid cells. DC migration to the local lymphoid tissues, as part of the adaptive immune response, is regulated and controlled by chemokines from other immune cells.
Some types of dendritic cells reside in the local lymphoid tissues and are thought to replenish immature populations, to transport self or environmental antigens within these tissues, or to be on patrol to identify invaders. (17, 18)
Monocytes
Monocytes are recruited from the circulation to the local area following viral infection as part of the inflammation process. They produce type I interferons (IFNs) and chemokines and can differentiate into macrophages or dendritic cells. By increasing the number and activity of these two cell types, both viral clearance and cytotoxic activity is amplified. However, these effects also involve additional stimulation of pro-inflammatory activity. Therefore, an excessive monocyte response may lead to inflammatory and immune-mediated damage, particularly when the infection has spreads into the lungs. Thus, the role of inflammatory monocytes in lung pathology may be detrimental or beneficial depending on the degree of the response and viral pathogen. (17)
Neutrophil leukocytes
The activity of neutrophils is one of the critical components in the progression and course of the acute viral URTI and represent an important component of the innate immune response. They are able to phagocytose virions (whole live viruses), viral particles, and apoptotic cell bodies containing the virus. Once phagocytosed, pathogens are killed or inactivated by proteolytic enzymes and antimicrobial peptides released from intracellular granules, as well as by internally produced reactive oxygen species (ROS). The contents of neutrophil granules as well as ROS may also be secreted and released extracellularly, including antimicrobial peptides as well as extracellular traps (NETs) that immobilize pathogens and prevent further dissemination. Thus, when recruited in sufficient numbers, activated neutrophils control virus propagation, and reduce disease severity. However, an excessive neutrophil response can be detrimental to the host by causing uncontrolled inflammation (particularly tissue swelling) and indiscriminate damage to both respiratory epithelium and the microvascular endothelium due to the pro-inflammatory and cytotoxic nature of the products that are released. (17)
Lymphocyte responses: from innate to adaptive
The second-tier immune defences are mediated by local tissue-resident lymphocytes. These include innate lymphoid cells (ILCs), natural killer (NK) cells, and tissue-resident memory T and B cells. These cells serve as an intermediary controller, responding to the cytokine signals from local sensor cells and producing effector cytokines that recruit specific subsets of effector cells to clear pathogens. These cells are already present at the time of infection and are thus distinct from recruited lymphocytes (T and B cells) that arrive as a consequence of adaptive immunity several days later.
The complement system
Complement is a major element of the innate immune response and also serves to link innate and adaptive immunity. Complement comprises over 30 serum and membrane-bound proteins which, when activated, form a cascade of reactions contributing to the elimination of invading microorganisms. The complement system functions to identify and opsonize (coat and inactivate) bacteria and other pathogens. It renders pathogens susceptible to phagocytosis and also directly kills some pathogens and infected cells. It promotes clearance of dead cells or antibody complexes and removes foreign substances present in organs, tissues, blood and lymph. It can also play a part in the activation of the adaptive immune response through the mobilization and activation of antigen-presenting cells.
The adaptive immune response
Although the innate immune response, as discussed above, plays a significant role in host antiviral responses, it mainly serves to slow rather than stop the infection. The adaptive immune response is generally responsible for complete viral clearance by halting viral replication to prevent the generation of new virions and eliminating infectious virions. The primary functions of the adaptive immune response are:
recognition of specific non-self antigens and distinguishing them from self antigens
generation of pathogen-specific immunologic effector pathways that eliminate specific pathogens or pathogen-infected cells
development of an immunologic memory that can quickly eliminate a specific pathogen in future infections with the same pathogen
Antigen presenting cells, mostly dendritic cells, migrate from the initial site of infection to the local lymphoid tissues (adenoids and tonsils) to prime the T-lymphocyte (T-cell) responses. This process activates two distinct pathways, leading on the one hand, to the generation of cytotoxic T cells that destroy host cells infected by the virus and, on the other, to the generation of specialised B-lymphocytes (B cells), which then change into antibody secreting plasma cells at the site of infection. Both cytotoxic and antibody secreting cells are specific for the infecting pathogen. The pathogen-specific lymphocytes migrate back to the site of infection, where the B cells are able to directly ‘recognise’ antigenic viral molecules, which causes them to proliferate and transform into antibody-secreting plasma cells. The plasma cells produce specific antibody, immunoglobulin A (IgA) type, that disables the virus, preventing entry into host cells. Antibody activates phagocytic cells and facilitates their activity, while also activating the complement system to induce extracellular destruction of inactivated virions. Antibodies are also able to ‘tag’ infected cells, marking them for destruction through apoptosis. Upon elimination of the pathogen, the antigen-antibody complexes are cleared by the complement system. As all viruses replicate within cells and many of them spread directly between cells without re-entering the extracellular environment, resolution of infection is reliant more on cytotoxic T-cell function than on antibody. (17, 18)
The cytotoxic activities of specialised T lymphocytes comprise the ‘cellular’ component of the adaptive immune response. By destroying infected cells, the cytotoxic T lymphocytes are essential in the complete resolution of viral infections. While most of the specialised T cells and plasma cells are only short lived, a small subset of these specialised T and B cells remains in the local tissues to provide immune memory in future encounters with the same pathogen. (17, 18)
Although cytotoxic activity of the immune system is essential to viral clearance, if cytotoxic processes within the lungs are excessive, respiratory function may be compromised through the loss of airway epithelium, disruption of the epithelial-endothelial barrier, and accumulation of apoptotic cells and cellular debris in the airways. This leads to respiratory distress syndrome, which is a medical emergency. (17, 18)
Viral infection and the immune response – summary
When a virus reaches the apical surface of the nasal epithelial cell layer, it is sensed by local macrophages, dendritic cells and epithelial cells. These sensor cells secrete cytokines and chemokines, which initiate a local innate immune response, mutually activating each other in the first step of a multistage response.
According to the current model, if pathogen clearance is not accomplished by these cells directly, then they attract neutrophils from the local blood vessels by chemotaxis (through secretion of chemokines) and activation of an inflammatory response, via pro-inflammatory cytokines, which dilates the local blood vessels, making them more permeable, and instructs the vascular endothelial cells to ‘grab onto’ circulating neutrophils. Other cytokines released by these sensor cells activate the phagocytic and cytotoxic activities of the newly recruited neutrophils.
If this step fails to contain and eliminate the virus, the sensor cells send chemical signals (via first-order cytokines) to the local resident immune cells (including killer T cells, memory T and B cells, natural killer cells and innate lymphoid cells), activating them to destroy the foreign invader as well as infected cells. This step is aimed at pathogen clearance, by phagocytosis, lysis, or binding of microorganisms, as well as the eventual activation of specific responses (i.e. adaptive immunity) if the virus has managed to escape the innate immune response.
The final stage of the innate immune response is activated if the resident immune cells are not able to overcome the infection on their own. In this case, they secrete specific second-order cytokines, which activate select effector cells to assist in this process. These are mostly immune cells (neutrophils, monocytes and macrophages), which are given additional stimulus to complete the process of pathogen clearance and promote repair of damaged epithelial tissue.
If successful viral clearance is achieved at any one of the above steps, intercellular signalling is damped down, most of the activated cells die and are cleared away along with other cellular debris, and damaged tissues are restored. This initial process is referred to as the innate immune response and it is a general response to an invading pathogen. It is more likely to be successful if some of the local memory cells have had previous contact with the specific virus and are can therefore be activated to mount a specific response to supplement the innate response.
If the virus manages to escape the innate immune mechanisms, antigen presenting cells (dendritic cells and macrophages) are chemically instructed to migrate into the neighbouring lymphoid tissues (adenoids and tonsils) and activate specific T cell and B cell lines. These new colonies of specific T cells are able to recognise host cells that are infected with the virus and destroy them; while the specialised B cells differentiate into plasma cells, which secrete a specific IgA antibody against the virus. These lines of activated specific cells migrate back to the local area to resolve the infection. This process may take several days. Therefore, if the virus is particularly virulent and the infection progresses rapidly, it may spread proximally into the lungs before the adaptive immune response is able to contain it. Moreover, in hosts who have had some of the local lymphoid tissue removed (e.g. by tonsillectomy or adenoidectomy) their adaptive immune response to a viral URTI is likely to be impaired, leaving them more susceptible to respiratory infections as well as allergic disease. (19)
Prolonged or excessive immune responses (both innate and adaptive) with failure of self-regulation may lead to cytokine storm, in which unregulated cytotoxic activity and inflammatory swelling lead to lung tissue damage and development of acute respiratory distress syndrome.
Inflammation
The symptomatology of AVR reflects the wide variety of host responses, which depend on the local immune response, the level of exposure to the pathogen and, to a much lesser extent, the unique features of the pathogenic virus itself. Patient age, overall health and co-morbidities also influence the symptomatology. Thus, while most viral URTI’s are associated with an acute self-limiting illness, depending on these other factors, they may also occur without symptoms, or they may kill. (13, 17)
This is an important concept: the severity, and indeed the lethality, of a viral URTI is mainly dependant on the host’s immune response to the infection. Of course, viral tropism, i.e. whether or not the infection tends to spread rapidly into the lungs, is an important factor. However, the major cause of severe and lethal respiratory distress syndrome is an excessive inflammatory response.
Symptomatology
The symptoms of AVR are triggered by the inflammatory cascade of the local immune system. The most significant signs of viral respiratory tract infections include sneezing, rhinorrhea (runny nose and nasal discharge), nasal congestion, cough, tachypnoea and fever. Subjective symptoms include a sore throat, malaise, shivering (chills), shortness of breath, muscle aches and weakness, fatigue, mood changes, loss of appetite and headaches. (13, 20)
Symptoms of upper respiratory tract infections have been traditionally classified as early or late. The early symptoms are those that develop quickly and resolve rapidly after one or two days, principally headaches, sneezing, chills, sore throat, and malaise. Late symptoms include nasal discharge, nasal obstruction, and cough. Generally, the severity of symptoms increases rapidly, peaking 2 – 3 days after infection, with a mean duration of 7 – 10 days but with some symptoms persisting for more than 3 weeks. The best predictors for influenza are cough and fever. (13, 20)
The immune response to infection is generally the main factor in causing the symptoms in a viral URTI, rather than damage to the airway. The initial cellular response to viral infiltration and infection is the production and release of cytokines and chemokines, which act to recruit other immune cells, trigger inflammation, and generate systemic symptoms such as fever. A complex mix of proinflammatory cytokines and mediators of inflammation gives rise to the symptoms in an URTI, as described below. (13, 20)
Sore throat
A sore throat is often the first symptom and is most likely caused by the actions of prostaglandins and bradykinin (both being produced due to the effects of pro-inflammatory cytokines) on sensory nerve endings in the airway. The sensation of pain is mediated by the cranial nerves supplying the nasopharynx and pharynx (vagus, glossopharyngeal and maxillary nerves). (13, 20)
Nasal congestion
Nasal congestion is due to dilation of the venous sinuses in the nasal epithelium in response to the vasodilator mediators, such as bradykinin. (13, 20)
Nasal discharge
This material is made up of a complex mixture of plasma and glandular exudates with cellular elements, such as goblet cells, plasma cells, and neutrophils. In the early phase, local branches of the trigeminal nerve are stimulated to dramatically increase glandular secretion. The concentrated mucins that are secreted into the lumen then draw in water from the dilated blood vessels by osmosis to swell to several hundred times their original dehydrated volume. The colour of the nasal discharge may change from watery clear to yellow and green during the course of the infection, and this reflects the severity of the inflammatory response rather than the aetiology of the infection. There is no evidence to support the idea that a green or yellow colour is a clinical marker of bacterial superinfection. The colour change is related to the recruitment of leukocytes into the airway lumen. Neutrophils and activated monocytes contain chromatic, green granules (azurophil granules). Thus, the more leukocytes present in the discharge the more colourful it appears. (13, 20)
Sneezing
Sneezing is mediated solely by the trigeminal nerves, which supply the nasal epithelium and the anterior part of the nasopharynx with sensory fibres. Sneezing is related to inflammatory responses in the nose and nasopharynx that stimulate the trigeminal nerves. The sneeze response may be mediated via histamine receptors on the trigeminal nerves. (13, 20)
Cough
Coughing is mediated by the vagus nerve and is an indication that the infection has spread below the nasopharynx. As the inflammatory process spreads proximally the sensory receptors of the vagus nerve are stimulated by inflammatory mediators. Coughing in an acute viral URTI is an indicator of significant damage to the respiratory epithelium proximal to the nasopharynx and is generally indicative of infection by influenza or influenza-like viruses. (13, 20)
Fever and chills
Fever arises as a result of the effects of cytokines that are released in response to a novel or severe viral infection. In children it may be induced by any of the viruses associated with acute URTI’s; however, in adults it is generally a response to influenza viruses. Chills or a fall in body temperature is a common symptom in adults. Both fever and chills are deemed to arise from the effects of cytokines on the temperature control centre in the hypothalamus. (13, 20)
General malaise and mood changes
Both malaise and low mood are thought to be caused by the effects of cytokines on the central nervous system. From an evolutionary perspective, both symptoms would predispose a person to rest and self-isolate, thus enhancing survival of both self and species. Anorexia is thought to be due to the action of cytokines on the feeding centre in the hypothalamus. (13, 20)
Headache
Headaches are most likely due to the action of cytokines and chemokines on local nociceptive sensory neurones. (20, 21)
Muscle pain
Myalgia during an acute URTI is thought to be due to the direct effects of pro-inflammatory cytokines (such as TNF-alpha) on muscle tissue as well as the effects of prostaglandin E2 (PGE2), which is produced in response to cytokines. Some pro-inflammatory cytokines have been shown to cause breakdown of skeletal muscle tissue and PGE2 is a known pain mediator. (20, 22)
Western medical interventions: the role of vaccines
In light of the above exposition, it should be obvious that a ‘shot-in-the-arm’ type medial intervention (with only systemic and not local, activity) will have little effect on the development of sterilising local immunity to a viral infection within the respiratory mucosa. Nor will a weak and rapidly waning local antibody response have much effect in assisting the host immune response to the infection. The infection will generally become established and persist, despite the intervention. Moreover, the rate at which viruses can mutate virtually guarantees the rapid obsolescence of an intervention that targets one specific virus only.
When antigenic peptides are injected intramuscularly (IM), they find their way, mainly via the draining lymph vessels, into the local lymph nodes. The B cells that reside there can respond to antigens directly, without requiring the intermediary of either an antigen-presenting cell (dendritic cell or macrophage) or a T-helper cell. These B cells are activated to proliferate and produce a specific antibody. However, this antibody is composed mostly of IgG (together with some IgM), which differs significantly from the mucosal IgA antibodies produced in the natural adaptive immune response. The addition of an adjuvant to a vaccine (e.g. an aluminium salt, oil-in-water emulsion, graphene oxide or lipid nanoparticles) allows activation of dendritic cells (DC) in the same way as they would respond to venoms and other toxic substances. (22) This sets off an entirely different immune pathway from that which would normally be triggered by a viral infection. DC’s take in and break down the antigen-adjuvant complex and present parts of the antigen and/or adjuvant to T cells within the local lymph nodes. However, this only induces a response from T-helper cells that will enhance the antibody production pathway; it does not lead to the production of antigen-specific cytotoxic T cells. Moreover, in both cases the initial site of ‘infection’ is the local muscle – not the respiratory tract. Therefore, chemotactic signalling will only direct the adaptive immune responses back to the original injection site, which is the main locus of inflammation.
Similarly, small amounts of antigen-adjuvant complex will find their way into the systemic circulation, where smaller amounts end up in lymphoid tissue disseminated throughout the body. However, without the elaborate chemical signalling that occurs between a local site of infection and its local draining lymph nodes, the adaptive immune response is not specifically directed to the respiratory mucosa. Even though ‘antibodies’ can be detected in the blood, this means very little in terms of mucosal immunity within the nose. Hopefully, advancing knowledge of the immune system may lead to more effective interventions in the future. (24, 25, 26)
The limitations of current IM delivered interventions for viral respiratory infections may be summarised as follows:
They produce only an humoral (i.e. antibody) response, when complete viral clearance requires both cellular (i.e. cytotoxic T-cells) and humoral adaptive immune responses
The type of antibody produced (mostly IgG) is not the correct type and has a much weaker effect than IgA that is produced through natural immunity. Additionally, IgG is pro-inflammatory in the lungs and may exacerbate lung damage and respiratory distress syndrome if the infection spreads into the lungs. (27)
The adaptive immune response to an IM injection is centred on the axillary lymph nodes and systemic circulation, when it needs to occur within the mucosa of the nose and other parts of the respiratory system.
In the words of a recent review paper: ‘… there exists an immunologic disconnect between some current vaccinations delivered systemically and protection afforded at mucosal barriers.’ (26) In other words, the real-world results of IM delivered vaccines for viral respiratory infections fall far short of those that were intended. Sadly, it appears that wishful thinking based upon plausibility and the measurement of surrogate markers has supplanted critical thinking and scientific evidence in the public discourse regarding vaccination against respiratory viral infections.
REFERENCES
1. Gotszche, P. (2020). The coronavirus pandemic: can we handle such epidemics better? J R Soc Med. 113(5): 171-5
2. Campbell, J. (2022), Freedom of information discussion. Dr Campbell YouTube Channel. Retrieved October, 2022 from:
3. Axfors, C., Ioannidis, J. (2022). Infection fatality rate of COVID-19 in community-dwelling elderly populations. Eur J Epidemiol. 37(3):235-249.
4. Compton, K., Connolly, K., Gerberich, A., (2022). Voixx. Drugwatch website. Retrieved, October, 2022, from: https://www.drugwatch.com/vioxx/
5. Gøtzsche P. (2021). Non-steroidal anti-inflammatory drugs do not have anti-inflammatory effects. Institute for Scientific Freedom. Retreived October, 2022 from: https://www.scientificfreedom.dk/2021/06/23/non-steroidal-anti-inflammatory-drugs-do-not-have-anti-inflammatory-effects/
6. Li, H., Li, Y., Luo, C., Liang, X., Liu, Z., Liu, Y., Ling, Y., (2022). New Approach for Targeted Treatment of Mild COVID-19 by Honeysuckle through Network Pharmacology Analysis. Comput Math Methods Med. 2022:9604456.
7. Çatlı, T., Atilla, H., Miller, E., (2020). Acute Viral Rhinitis. In: Cingi, C., Bayar Muluk, N. (eds) All Around the Nose. Springer, Cham.
8. Kuchar, E., Miśkiewicz, K., Nitsch-Osuch, A., Szenborn, L., (2015). Pathophysiology of Clinical Symptoms in Acute Viral Respiratory Tract Infections. Adv Exp Med Biol.857:25-38.
9. Zanotti, G., Grinzato, A., (2021). Structure of filamentous viruses. Curr Opin Virol. 51:25-33.
10. Praena, B., Wan, X.,(2022). Influenza Virus Infections in Polarized Cells. Viruses. 14(6):1307.
11. Michael D Vahey, M., Fletcher, D. (2019). Influenza A virus surface proteins are organized to help penetrate host mucus. eLife 8:e43764.
12. Kaler, L., Iverson, E., Bader, S., Song, D., Scull, M., Duncan, G. (2022). Influenza A virus diffusion through mucus gel networks. Commun Biol. 5(1):249.
13. Eccles, R. (2005). Understanding the symptoms of the common cold and influenza. Lancet Infect Dis. 5(11):718-25.
14. Dimitri-Pinheiro, S., Soares, R., Barata, P. (2020). The Microbiome of the Nose-Friend or Foe? Allergy Rhinol (Providence). 11:2152656720911605.
15. Hanada, S., Pirzadeh, M., Carver, K., Deng, J. (2018). Respiratory Viral Infection-Induced Microbiome Alterations and Secondary Bacterial Pneumonia. Front Immunol. 9:2640.
16. Iwasaki, A., Foxman, E., Molony, R. (2017). Early local immune defences in the respiratory tract. Nat Rev Immunol. 17(1):7-20.
17. Newton, A., Cardani, A., Braciale, T. (2016). The host immune response in respiratory virus infection: balancing virus clearance and immunopathology. Semin Immunopathol. 38(4):471-82.
18. Mueller, S., Rouse, B., (2008). Immune responses to viruses. Clinical Immunology. 2008:421–31.
19. Byars, S., Stearns, S., Boomsma, J., (2018). Association of Long-Term Risk of Respiratory, Allergic, and Infectious Diseases With Removal of Adenoids and Tonsils in Childhood. JAMA Otolaryngol Head Neck Surg.144(7):594-603.
20. Kuchar, E., Miśkiewicz, K., Nitsch-Osuch, A., Szenborn, L. (2015). Pathophysiology of Clinical Symptoms in Acute Viral Respiratory Tract Infections. Adv Exp Med Biol. 857:25-38.
21. Tolebeyan, A., Zhang, N., Cooper, V., Kuruvilla, D. (2020). Headache in Patients With Severe Acute Respiratory Syndrome Coronavirus 2 Infection: A Narrative Review. Headache. 60(10):2131-2138.
22. Widyadharma, I., Dewi, P., Wijayanti, I., Utami, D. (2020). Pain related viral infections: a literature review. Egypt J Neurol Psychiatr Neurosurg. 2020;56(1):105.
23. Ryan, R., Seymour, J., Loukas, A., Lopez, J., Ikonomopoulou, M., Miles, J., (2021). Immunological Responses to Envenomation. Front Immunol. 12:661082.
24. Clem AS. Fundamentals of vaccine immunology. J Glob Infect Dis. 2011 Jan;3(1):73-8.
25. Pollard AJ, Bijker EM. A guide to vaccinology: from basic principles to new developments. Nat Rev Immunol. 2021 Feb;21(2):83-100.
26. Mettelman RC, Allen EK, Thomas PG. Mucosal immune responses to infection and vaccination in the respiratory tract. Immunity. 2022 May 10;55(5):749-780.
27. Russell MW, Moldoveanu Z, Ogra PL, Mestecky J. Mucosal Immunity in COVID-19: A Neglected but Critical Aspect of SARS-CoV-2 Infection. Front Immunol. 2020 Nov 30;11:611337.